|
Quantity
|
Out of stock
|
||
|
|
|||
Instructions for use of Biokan DHPPi+LR vaccine
for the prevention of plague, adenovirus, infectious hepatitis, parvovirus, parainfluenza, leptospirosis and canine rabies
(Developer organization: JSC Bioveta, Czech Republic)
I. General information
Trade name: vaccine Biokan DHPPi+LR.
International non-proprietary name: vaccine for the prevention of plague, adenovirus, infectious hepatitis, parvovirus, parainfluenza, leptospirosis and canine rabies.
Pharmaceutical form: dry component Biokan DHPPi (lyophilized mass) and liquid component Biokan LR – suspension for injections.
The dry component is made from a culture suspension of cells of the VERO line. infected with attenuated canine plague viruses (strain CDVU 39) and canine parainfluenza (strain CPIV-2); culture suspension of MDCK cells infected with attenuated adenovirus of dogs (CAV-2 strain); culture suspension of CRFK cells infected with attenuated canine parvovirus (CPV OR strain - 1/81), with the addition of auxiliary substances (trometamol, helaton, sucrose, dextran) 30%.
The liquid component is made from inactivated rabies virus (Vnukovo-32 strain), cultivated on the VNK-21 cell line, and inactivated leptospiral serogroups L. icterohaemorrhagiae, L. canicola, L. grippotyphosa with the addition of auxiliary substances: 10%.
The dry component looks like a dry porous mass of cream or pink color, the liquid component is a pink-red liquid with a sediment, when shaken, it easily breaks into a homogeneous suspension.
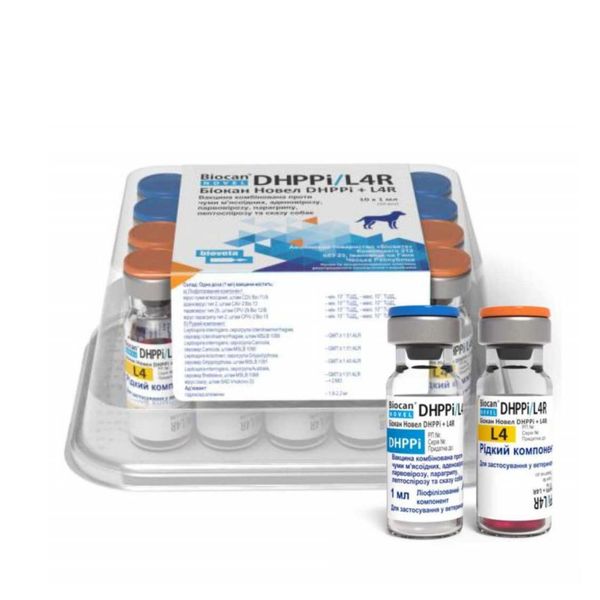
The components of the vaccine are packaged in glass vials of 1.0 ml (one immunizing dose).
Vials are hermetically sealed with rubber stoppers, reinforced with aluminum caps.
Vaccine vials are packed in plastic containers: 5 immunization doses (10 vials), 10 immunization doses (20 vials), 50 immunization doses (100 vials).
Instructions for use are included in each box with the vaccine.
The shelf life of the vaccine is 24 months from the date of issue, subject to compliance with storage and transportation conditions.
After the expiration date, the vaccine is unusable.
The vaccine is stored and transported in the manufacturer's closed packaging in a dry, light-protected place at a temperature of 2 to 8°C, separately from food and feed.
The vaccine should be stored in a place inaccessible to children.
Vials with vaccine without labels, with an expired expiration date, with a violation of the integrity and/or tightness of the closure, with a changed color and/or consistency of the contents, with the presence of foreign impurities, as well as vaccine residues not used within 4 hours after opening the vials, culling and disinfection by boiling for 30 minutes or treatment with 2% alkali solution or 5% chloramine solution (1:1) for 30 minutes.
Disposing of the decontaminated vaccine does not require special precautions.
II. Biological properties
The Biokan DHPPi+LR vaccine causes the formation of an immune response in dogs to the causative agents of plague, adenovirus, infectious hepatitis, parvovirus, parainfluenza, leptospirosis and rabies 14-21 days after revaccination, which persists for at least 12 months.
One immunizing dose contains: at least 103.0 TCID50 of attenuated carnivore plague virus; not less than 103.5 TCID50 of attenuated adenovirus of dogs; not less than 104.5 TCID50 of attenuated canine parvovirus; not less than 103.0 TCID50 of attenuated canine parainfluenza virus; inactivated L. icterohaemorrhagiae, L. canicola, L. grippotyphosa; not less than 1 IU/ml of inactivated rabies virus.
The vaccine is harmless and has no medicinal properties.
III. Application procedure
The Biokan DHPPi+LR vaccine is intended for the prevention of plague, adenovirus, infectious hepatitis, parvovirus, parainfluenza, leptospirosis and rabies in dogs.
It is forbidden to vaccinate clinically sick and/or weakened animals.
Preventive deworming should be carried out 10 days before immunization.
It is forbidden to use the drug in pregnant and lactating dogs.
Dogs are subject to vaccination starting from 8-10 weeks of age.
The vaccine is injected subcutaneously in a dose of 1 ml, regardless of the breed and weight of the animal.
Primary vaccination is carried out at the age of 8-10 weeks, followed by revaccination after 3-4 weeks, but not earlier than 12 weeks of age.
In the future, animals are vaccinated once a year.
Immediately before use, dry and liquid components are combined in one bottle.
Vaccination is carried out in compliance with the rules of asepsis and antiseptics.
No symptoms of plague, adenovirosis, infectious hepatitis, parvovirus, parainfluenza, leptospirosis and rabies or other pathological signs of vaccine overdose have been established.
Features of the post-vaccination reaction during primary and repeated immunizations have not been established.
Violation of the scheme (terms) of vaccine administration should be avoided, as this may lead to a decrease in the effectiveness of immunopro. lactic plague, adenovirus, infectious hepatitis, parvovirus, parainfluenza, leptospirosis and rabies.
In case of missing the next injection of the vaccine, it is necessary to carry out the immunization as soon as possible.
When using the vaccine in accordance with this instruction, side effects and complications, such as pusually not noted.
It is forbidden to use Biokan DHPPi+LR vaccine simultaneously with other immunobiological drugs.
The use of other immunobiological drugs is possible with a 14-day interval before or after the introduction of the Biokan DHPPi+LR vaccine.
The terms of use of products of animal origin after the introduction of the Biokan DHPPi+LR vaccine are not established.
IV. Personal prevention measures
When working with the Biokan DHPPi+LR vaccine, you should follow the general rules of personal hygiene and safety techniques provided for when working with medicinal products.
All persons participating in vaccination must be in overalls and equipped with personal protective equipment. There must be a first aid kit in the workplace.
In case of accidental contact of the medicinal product with the skin or mucous membranes, they must be washed with a large amount of clean water. In case of vaccine spillage, contaminated surfaces should be treated with 2% alkali solution or 5% chloramine solution, tools should be decontaminated by boiling for 30 minutes.
If the drug is accidentally administered to a person, the injection site must be immediately treated with 70% ethyl alcohol, contact a medical facility and inform the doctor about it.